Differences Between Qualitative and Quantitative Research (And When to Use Both)
Both qualitative and quantitative research help inform important decisions throughout product development.
Both qualitative and quantitative research help inform important decisions throughout product development.
Collaboration between Human Factors Engineers and Designers is crucial for cultivating an understanding of individual goals and working together to meet those goals in product development.
Our team has researched users and developed products for markets all over the world. Learn more about our global perspective:
Continuing the conversation on Design Strategy with a look into our Strategic Concept Vision. Learn how we help our clients evaluate and build upon a prospective future.
What is Design Strategy at Insight Product Development? We’re sharing how we provide value to our clients by leveraging this stage of innovation.
Gaining access to facilities and participants for ethnographic research has always been a significant challenge for our clients. Learn how our research team employs a multi-faceted approach to overcoming recruiting challenges to inform medical device development.
Learn how we utilize iterative prototyping to effectively push risk upstream and the value that each stage of prototyping provides as designs transition from engineering into manufacturing.
Sometimes it’s not all that easy to identify and prioritize user needs to ensure that they drive relevant decisions throughout development. Check out our Q&A with Insight’s Director of Research and Strategy Carolyn Rose to get some insights on this topic.
 Craig Scherer and Carolyn Rose talk about the importance of effectively identifying, prioritizing, and translating user needs into design guidance in their latest Med Device Online article
Craig Scherer and Carolyn Rose talk about the importance of effectively identifying, prioritizing, and translating user needs into design guidance in their latest Med Device Online article Turning user needs into design guidance is a key component of developing a successful medical device. The primary goal of this process is to develop a device that not only meets the FDA’s criteria for safety and effectiveness, but also one that provides the best possible user experience for all stakeholders.
Turning user needs into design guidance is a key component of developing a successful medical device. The primary goal of this process is to develop a device that not only meets the FDA’s criteria for safety and effectiveness, but also one that provides the best possible user experience for all stakeholders.
The successful application of user-centered design principles relies on iterative and frequent feedback from stakeholders throughout the entire development process. This research also provides the design team information with which to identify, document, and better manage risk throughout the design cycle.
Learn more about these six tools in the full article on MedDevice Online.
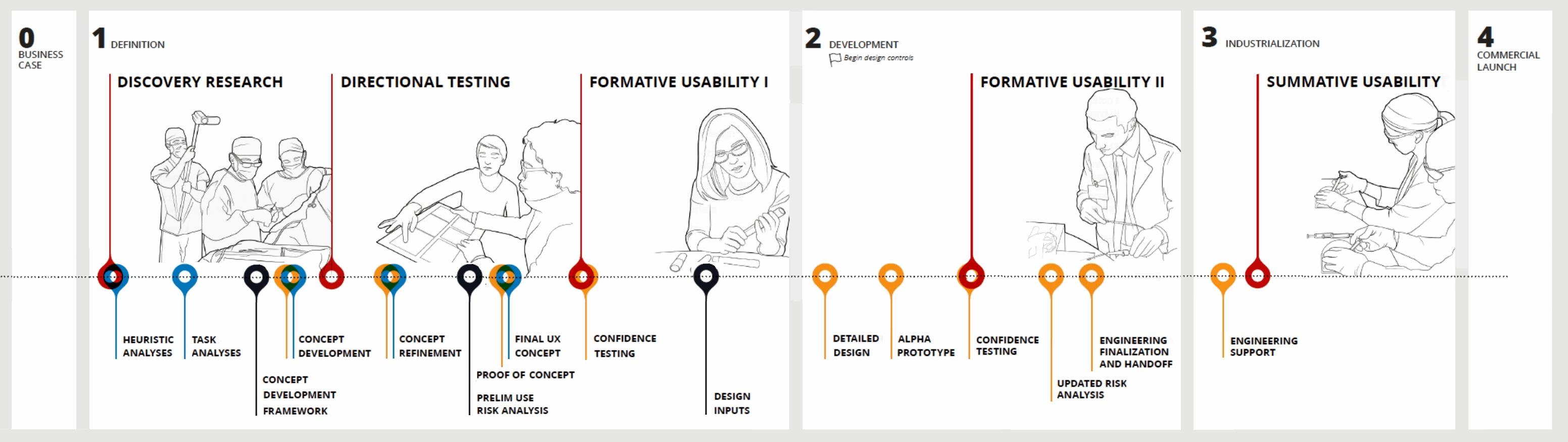
For a deeper dive in our user research activities, check out our user needs poster.
Effectively identifying, prioritizing and translating user needs into design guidance is critical to successfully developing a safe, effective and desirable medical device.
