MX Magazine: Technology, Start Your Engine
 Leveraging technology in the right way is critical to making progress in product development. As a medical product developer technology-related concerns raise many questions, but two fundamental ones stand out:
Leveraging technology in the right way is critical to making progress in product development. As a medical product developer technology-related concerns raise many questions, but two fundamental ones stand out:
- What can a company do with its technology?
- How can a company use technology to make its idea real?

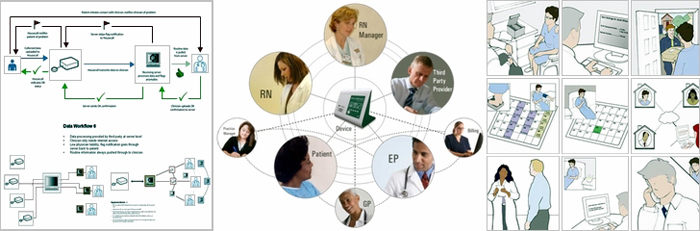
 AAMI’s HE75 and the new FDA draft guidance, “Applying Human Factors and Usability Engineering to Optimize Medical Device Design” has officially established human factors activities as a requirement throughout the development process, leading many to ask if human factors is something that their company knows how to do, has to do or can do. Because the FDA has reaffirmed the perspective that human factors is a critical dimension of safe and effective device design, human factors and usability must be considered from the outset and their ultimate verification and validation are rooted in the very same regulatory basis as technical device function.
AAMI’s HE75 and the new FDA draft guidance, “Applying Human Factors and Usability Engineering to Optimize Medical Device Design” has officially established human factors activities as a requirement throughout the development process, leading many to ask if human factors is something that their company knows how to do, has to do or can do. Because the FDA has reaffirmed the perspective that human factors is a critical dimension of safe and effective device design, human factors and usability must be considered from the outset and their ultimate verification and validation are rooted in the very same regulatory basis as technical device function.  On June 22, 2011, the FDA released a new draft guidance, “Applying Human Factors and Usability Engineering to Optimize Medical Device Design.” This guidance has implications for all medical device manufacturers. Once this draft is finalized it will set a new standard for integrating HF practices within the development process, and provide a more transparent understanding of what HF considerations the FDA will be looking for when evaluating a device submission.
On June 22, 2011, the FDA released a new draft guidance, “Applying Human Factors and Usability Engineering to Optimize Medical Device Design.” This guidance has implications for all medical device manufacturers. Once this draft is finalized it will set a new standard for integrating HF practices within the development process, and provide a more transparent understanding of what HF considerations the FDA will be looking for when evaluating a device submission.